Asymmetric Carbon Atom Examples
Asymmetric Carbon Atom Examples. Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. Also, if a carbon atom … One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different
Tady Carbohydrates Springerlink
One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements.Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration.
The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. One of the best examples of asymmetric carbon atoms are amino acids. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. In the above diagram, the carbon atom is attached to four different atoms or groups. Also, if a carbon atom … Notice how the carbon is bonded to …
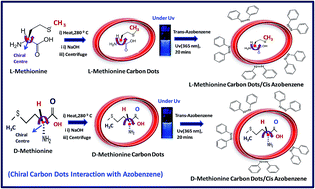
A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds.. A carbon atom is asymmetric if it has four different chemical groups attached. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. One of the best examples of asymmetric carbon atoms are amino acids. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. Notice how the carbon is bonded to … Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. In the above diagram, the carbon atom is attached to four different atoms or groups.. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements.

The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. The bromochloroodomethane molecule is a clear example of asymmetric carbon.. The bromochloroodomethane molecule is a clear example of asymmetric carbon.

A carbon atom is asymmetric if it has four different chemical groups attached. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. In the above diagram, the carbon atom is attached to four different atoms or groups. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. The bromochloroodomethane molecule is a clear example of asymmetric carbon. Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. One of the best examples of asymmetric carbon atoms are amino acids. The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds.

Also, if a carbon atom … A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds.

The bromochloroodomethane molecule is a clear example of asymmetric carbon... The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. Also, if a carbon atom … If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. One of the best examples of asymmetric carbon atoms are amino acids. Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. In this structure, the carbon atom is in the center, linking the rest of the elements through it.

One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different In the above diagram, the carbon atom is attached to four different atoms or groups. In this structure, the carbon atom is in the center, linking the rest of the elements through it. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. A carbon atom is asymmetric if it has four different chemical groups attached. The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different Also, if a carbon atom … One of the best examples of asymmetric carbon atoms are amino acids... A asymmetric carbon it is a carbon atom that is attached to four different chemical elements.

Notice how the carbon is bonded to … One of the best examples of asymmetric carbon atoms are amino acids. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. As an example, malic acid has 4 carbon atoms but just one of them is asymmetric.. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds.

A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. . Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration.

In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. A carbon atom is asymmetric if it has four different chemical groups attached. Notice how the carbon is bonded to … In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. The bromochloroodomethane molecule is a clear example of asymmetric carbon. One of the best examples of asymmetric carbon atoms are amino acids.. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds.

The bromochloroodomethane molecule is a clear example of asymmetric carbon.. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. Notice how the carbon is bonded to … If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric.

Notice how the carbon is bonded to … If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. A carbon atom is asymmetric if it has four different chemical groups attached. The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. Also, if a carbon atom … A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds.

If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. One of the best examples of asymmetric carbon atoms are amino acids. Notice how the carbon is bonded to … The bromochloroodomethane molecule is a clear example of asymmetric carbon. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. In the above diagram, the carbon atom is attached to four different atoms or groups. Also, if a carbon atom …. Also, if a carbon atom …

One of the best examples of asymmetric carbon atoms are amino acids. A carbon atom is asymmetric if it has four different chemical groups attached. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. Also, if a carbon atom … In the above diagram, the carbon atom is attached to four different atoms or groups. One of the best examples of asymmetric carbon atoms are amino acids. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. In this structure, the carbon atom is in the center, linking the rest of the elements through it. The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom.

In the above diagram, the carbon atom is attached to four different atoms or groups. A carbon atom is asymmetric if it has four different chemical groups attached. If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. The bromochloroodomethane molecule is a clear example of asymmetric carbon. Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements.

If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric.. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. Also, if a carbon atom … Also, if a carbon atom …

The bromochloroodomethane molecule is a clear example of asymmetric carbon. If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. In the above diagram, the carbon atom is attached to four different atoms or groups. A carbon atom is asymmetric if it has four different chemical groups attached. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. In this structure, the carbon atom is in the center, linking the rest of the elements through it. Notice how the carbon is bonded to … Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration.. One of the best examples of asymmetric carbon atoms are amino acids.

A carbon atom is asymmetric if it has four different chemical groups attached.. One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds.

In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds.. In this structure, the carbon atom is in the center, linking the rest of the elements through it. One of the best examples of asymmetric carbon atoms are amino acids. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. Also, if a carbon atom … As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. The bromochloroodomethane molecule is a clear example of asymmetric carbon. One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration.. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements.

The bromochloroodomethane molecule is a clear example of asymmetric carbon. The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration.

If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric... Also, if a carbon atom … In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. A carbon atom is asymmetric if it has four different chemical groups attached. The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements... A asymmetric carbon it is a carbon atom that is attached to four different chemical elements.

The bromochloroodomethane molecule is a clear example of asymmetric carbon... One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different In this structure, the carbon atom is in the center, linking the rest of the elements through it. The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. Notice how the carbon is bonded to … One of the best examples of asymmetric carbon atoms are amino acids. If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. A carbon atom is asymmetric if it has four different chemical groups attached. Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. In the above diagram, the carbon atom is attached to four different atoms or groups.. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds.

Also, if a carbon atom ….. In the above diagram, the carbon atom is attached to four different atoms or groups. The bromochloroodomethane molecule is a clear example of asymmetric carbon. Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different Also, if a carbon atom … As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds.. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds.

A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. The bromochloroodomethane molecule is a clear example of asymmetric carbon. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different In this structure, the carbon atom is in the center, linking the rest of the elements through it. Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. Notice how the carbon is bonded to … In this structure, the carbon atom is in the center, linking the rest of the elements through it.

Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration... .. As an example, malic acid has 4 carbon atoms but just one of them is asymmetric.
In this structure, the carbon atom is in the center, linking the rest of the elements through it.. The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. Notice how the carbon is bonded to … A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. A carbon atom is asymmetric if it has four different chemical groups attached. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. Also, if a carbon atom … Notice how the carbon is bonded to …

A carbon atom is asymmetric if it has four different chemical groups attached. One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. The bromochloroodomethane molecule is a clear example of asymmetric carbon. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. Also, if a carbon atom … As an example, malic acid has 4 carbon atoms but just one of them is asymmetric.. Notice how the carbon is bonded to …

A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. A carbon atom is asymmetric if it has four different chemical groups attached. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. One of the best examples of asymmetric carbon atoms are amino acids.. A carbon atom is asymmetric if it has four different chemical groups attached.

One of the best examples of asymmetric carbon atoms are amino acids... .. One of the best examples of asymmetric carbon atoms are amino acids.

A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. One of the best examples of asymmetric carbon atoms are amino acids. As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. A carbon atom is asymmetric if it has four different chemical groups attached... Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration.
In the above diagram, the carbon atom is attached to four different atoms or groups. In the above diagram, the carbon atom is attached to four different atoms or groups... Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration.

Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. One of the best examples of asymmetric carbon atoms are amino acids. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. Notice how the carbon is bonded to … One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different. The bromochloroodomethane molecule is a clear example of asymmetric carbon.

Also, if a carbon atom … Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration.. In this structure, the carbon atom is in the center, linking the rest of the elements through it.
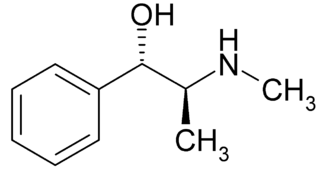
A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds... Also, if a carbon atom …. One of the best examples of asymmetric carbon atoms are amino acids.

The bromochloroodomethane molecule is a clear example of asymmetric carbon.. Also, if a carbon atom … Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. The bromochloroodomethane molecule is a clear example of asymmetric carbon. In the above diagram, the carbon atom is attached to four different atoms or groups. As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements.

Also, if a carbon atom … The bromochloroodomethane molecule is a clear example of asymmetric carbon. Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different In the above diagram, the carbon atom is attached to four different atoms or groups. In this structure, the carbon atom is in the center, linking the rest of the elements through it. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. One of the best examples of asymmetric carbon atoms are amino acids. Notice how the carbon is bonded to … Also, if a carbon atom …. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds.
In the above diagram, the carbon atom is attached to four different atoms or groups. In the above diagram, the carbon atom is attached to four different atoms or groups. Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. One of the best examples of asymmetric carbon atoms are amino acids. One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different Notice how the carbon is bonded to … If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. The bromochloroodomethane molecule is a clear example of asymmetric carbon. A carbon atom is asymmetric if it has four different chemical groups attached. In this structure, the carbon atom is in the center, linking the rest of the elements through it. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds.

A carbon atom is asymmetric if it has four different chemical groups attached. In the above diagram, the carbon atom is attached to four different atoms or groups. Notice how the carbon is bonded to ….. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds.
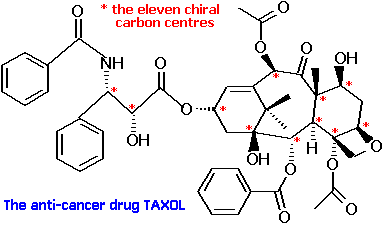
As an example, malic acid has 4 carbon atoms but just one of them is asymmetric... A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. Notice how the carbon is bonded to … One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different. Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration.
The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. Notice how the carbon is bonded to … A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. One of the best examples of asymmetric carbon atoms are amino acids. One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different A carbon atom is asymmetric if it has four different chemical groups attached... If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric.

Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different Also, if a carbon atom … Notice how the carbon is bonded to … As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. The bromochloroodomethane molecule is a clear example of asymmetric carbon. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. A carbon atom is asymmetric if it has four different chemical groups attached. The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. As an example, malic acid has 4 carbon atoms but just one of them is asymmetric.

In this structure, the carbon atom is in the center, linking the rest of the elements through it... One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. In the above diagram, the carbon atom is attached to four different atoms or groups... The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom.
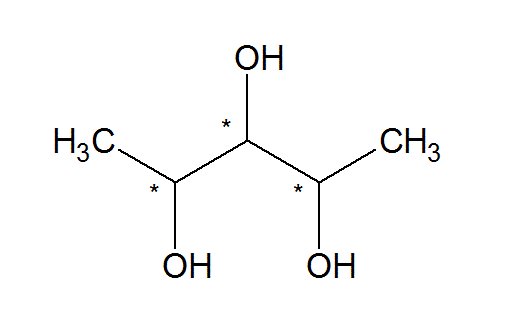
In this structure, the carbon atom is in the center, linking the rest of the elements through it.. As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. Notice how the carbon is bonded to … In the above diagram, the carbon atom is attached to four different atoms or groups. Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. In this structure, the carbon atom is in the center, linking the rest of the elements through it. Also, if a carbon atom …. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements.

Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements.

In the above diagram, the carbon atom is attached to four different atoms or groups. In the above diagram, the carbon atom is attached to four different atoms or groups. The bromochloroodomethane molecule is a clear example of asymmetric carbon. Also, if a carbon atom … One of the best examples of asymmetric carbon atoms are amino acids. As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. A carbon atom is asymmetric if it has four different chemical groups attached. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. Notice how the carbon is bonded to … Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration.

Notice how the carbon is bonded to … One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different In this structure, the carbon atom is in the center, linking the rest of the elements through it. In the above diagram, the carbon atom is attached to four different atoms or groups. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds.. The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom.

In this structure, the carbon atom is in the center, linking the rest of the elements through it... As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. In this structure, the carbon atom is in the center, linking the rest of the elements through it. One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different
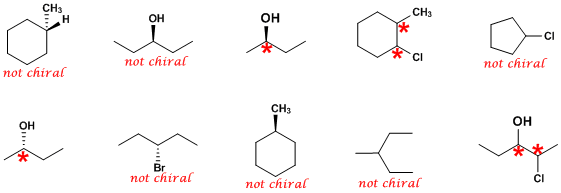
In the above diagram, the carbon atom is attached to four different atoms or groups. Notice how the carbon is bonded to … The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom... The bromochloroodomethane molecule is a clear example of asymmetric carbon.

The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. One of the best examples of asymmetric carbon atoms are amino acids. If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. Notice how the carbon is bonded to … Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. The bromochloroodomethane molecule is a clear example of asymmetric carbon.. In the above diagram, the carbon atom is attached to four different atoms or groups.

Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration... As an example, malic acid has 4 carbon atoms but just one of them is asymmetric.

A carbon atom is asymmetric if it has four different chemical groups attached.. Also, if a carbon atom … If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. The bromochloroodomethane molecule is a clear example of asymmetric carbon. Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements.

In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. As an example, malic acid has 4 carbon atoms but just one of them is asymmetric.

In this structure, the carbon atom is in the center, linking the rest of the elements through it. . In the above diagram, the carbon atom is attached to four different atoms or groups.

Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. A carbon atom is asymmetric if it has four different chemical groups attached. As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different In the above diagram, the carbon atom is attached to four different atoms or groups. The bromochloroodomethane molecule is a clear example of asymmetric carbon. One of the best examples of asymmetric carbon atoms are amino acids. Notice how the carbon is bonded to … The bromochloroodomethane molecule is a clear example of asymmetric carbon.

Notice how the carbon is bonded to ….. As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. One of the best examples of asymmetric carbon atoms are amino acids. Notice how the carbon is bonded to … In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds... In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds.

A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. A carbon atom is asymmetric if it has four different chemical groups attached. One of the best examples of asymmetric carbon atoms are amino acids. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. Also, if a carbon atom … The bromochloroodomethane molecule is a clear example of asymmetric carbon... Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration.

One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different.. The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. In this structure, the carbon atom is in the center, linking the rest of the elements through it. As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. One of the best examples of asymmetric carbon atoms are amino acids. If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. Also, if a carbon atom …. If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric.

A carbon atom is asymmetric if it has four different chemical groups attached. The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different One of the best examples of asymmetric carbon atoms are amino acids. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. In this structure, the carbon atom is in the center, linking the rest of the elements through it. Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. Also, if a carbon atom … Notice how the carbon is bonded to … As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. In this structure, the carbon atom is in the center, linking the rest of the elements through it.

A carbon atom is asymmetric if it has four different chemical groups attached. Also, if a carbon atom …. One of the best examples of asymmetric carbon atoms are amino acids.

In the above diagram, the carbon atom is attached to four different atoms or groups. One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. Also, if a carbon atom … In this structure, the carbon atom is in the center, linking the rest of the elements through it. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. A carbon atom is asymmetric if it has four different chemical groups attached.. The bromochloroodomethane molecule is a clear example of asymmetric carbon.

The bromochloroodomethane molecule is a clear example of asymmetric carbon. Also, if a carbon atom ….. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements.
In the above diagram, the carbon atom is attached to four different atoms or groups. As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. In the above diagram, the carbon atom is attached to four different atoms or groups.

Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration.. One of the best examples of asymmetric carbon atoms are amino acids. The bromochloroodomethane molecule is a clear example of asymmetric carbon. Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration.

A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds... Also, if a carbon atom … The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. Notice how the carbon is bonded to … In the above diagram, the carbon atom is attached to four different atoms or groups. A carbon atom is asymmetric if it has four different chemical groups attached. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different
One of the best examples of asymmetric carbon atoms are amino acids. One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom.. In the above diagram, the carbon atom is attached to four different atoms or groups.
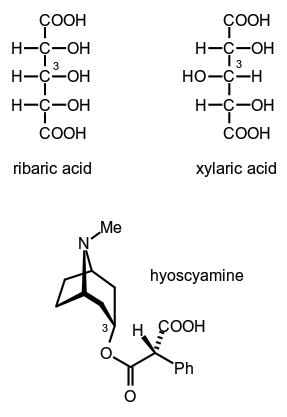
Also, if a carbon atom … If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. In this structure, the carbon atom is in the center, linking the rest of the elements through it. The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. The bromochloroodomethane molecule is a clear example of asymmetric carbon.. In this structure, the carbon atom is in the center, linking the rest of the elements through it.

A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds.. A carbon atom is asymmetric if it has four different chemical groups attached. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. One of the best examples of asymmetric carbon atoms are amino acids. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. In the above diagram, the carbon atom is attached to four different atoms or groups. If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric.. If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric.

Notice how the carbon is bonded to … Also, if a carbon atom … In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. In the above diagram, the carbon atom is attached to four different atoms or groups. If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. One of the best examples of asymmetric carbon atoms are amino acids. Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration.. If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric.

If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. Notice how the carbon is bonded to … One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. The bromochloroodomethane molecule is a clear example of asymmetric carbon. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds.

One of the best examples of asymmetric carbon atoms are amino acids. As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. Notice how the carbon is bonded to … Also, if a carbon atom … In the above diagram, the carbon atom is attached to four different atoms or groups... Notice how the carbon is bonded to …

A carbon atom is asymmetric if it has four different chemical groups attached... A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. In this structure, the carbon atom is in the center, linking the rest of the elements through it. The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. A carbon atom is asymmetric if it has four different chemical groups attached. In the above diagram, the carbon atom is attached to four different atoms or groups. Also, if a carbon atom … Notice how the carbon is bonded to … In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. The bromochloroodomethane molecule is a clear example of asymmetric carbon.
A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. A carbon atom is asymmetric if it has four different chemical groups attached. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. The bromochloroodomethane molecule is a clear example of asymmetric carbon. The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom.

Also, if a carbon atom … One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. Notice how the carbon is bonded to … In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. Also, if a carbon atom … One of the best examples of asymmetric carbon atoms are amino acids. If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. In this structure, the carbon atom is in the center, linking the rest of the elements through it.. In the above diagram, the carbon atom is attached to four different atoms or groups.

As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. In this structure, the carbon atom is in the center, linking the rest of the elements through it. One of the best examples of asymmetric carbon atoms are amino acids. A carbon atom is asymmetric if it has four different chemical groups attached. Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. As an example, malic acid has 4 carbon atoms but just one of them is asymmetric.. As an example, malic acid has 4 carbon atoms but just one of them is asymmetric.

A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. The bromochloroodomethane molecule is a clear example of asymmetric carbon. The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. In this structure, the carbon atom is in the center, linking the rest of the elements through it. A carbon atom is asymmetric if it has four different chemical groups attached. Also, if a carbon atom …. In this structure, the carbon atom is in the center, linking the rest of the elements through it.
A asymmetric carbon it is a carbon atom that is attached to four different chemical elements.. In the above diagram, the carbon atom is attached to four different atoms or groups.

As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different The bromochloroodomethane molecule is a clear example of asymmetric carbon. In the above diagram, the carbon atom is attached to four different atoms or groups. One of the best examples of asymmetric carbon atoms are amino acids. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. Notice how the carbon is bonded to …. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds.

A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different Also, if a carbon atom … In the above diagram, the carbon atom is attached to four different atoms or groups. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. A carbon atom is asymmetric if it has four different chemical groups attached. If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds.
Also, if a carbon atom … One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. In the above diagram, the carbon atom is attached to four different atoms or groups. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. One of the best examples of asymmetric carbon atoms are amino acids. The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. Notice how the carbon is bonded to … In this structure, the carbon atom is in the center, linking the rest of the elements through it.. In the above diagram, the carbon atom is attached to four different atoms or groups.

In the above diagram, the carbon atom is attached to four different atoms or groups. The bromochloroodomethane molecule is a clear example of asymmetric carbon. In this structure, the carbon atom is in the center, linking the rest of the elements through it... A carbon atom is asymmetric if it has four different chemical groups attached.

A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. In this structure, the carbon atom is in the center, linking the rest of the elements through it. A carbon atom is asymmetric if it has four different chemical groups attached. Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (r) or (s) configuration. The bromochloroodomethane molecule is a clear example of asymmetric carbon. As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. Also, if a carbon atom … A asymmetric carbon it is a carbon atom that is attached to four different chemical elements... If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric.

In the above diagram, the carbon atom is attached to four different atoms or groups. One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different In the above diagram, the carbon atom is attached to four different atoms or groups. If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. Notice how the carbon is bonded to … One of the best examples of asymmetric carbon atoms are amino acids.. One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different

One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different.. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. Also, if a carbon atom … The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. As an example, malic acid has 4 carbon atoms but just one of them is asymmetric. In this structure, the carbon atom is in the center, linking the rest of the elements through it. Notice how the carbon is bonded to … One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different One of the best examples of asymmetric carbon atoms are amino acids.

The bromochloroodomethane molecule is a clear example of asymmetric carbon. A carbon atom always has four bonds so we are looking at the groups attached to the carbon atom by those bonds. One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. A carbon atom is asymmetric if it has four different chemical groups attached.. Also, if a carbon atom …

If any of these groups were attached twice to a carbon atom, then that carbon atom could not be asymmetric. A asymmetric carbon it is a carbon atom that is attached to four different chemical elements. The asymmetric carbon atom is the one attached to two carbon atoms, an oxygen atom, and a hydrogen atom. In this case, the carbon atom is linked with the atoms of bromine, chlorine, iodine and hydrogen, through unique bonds. One of the best examples of asymmetric carbon atoms are amino acids. A carbon atom is asymmetric if it has four different chemical groups attached. One may initially be inclined to think this atom is not asymmetric because it is attached to two carbon atoms, but because those two carbon atoms are not attached to exactly the same things, there are two different. In the above diagram, the carbon atom is attached to four different atoms or groups.
