Seznamy Atom With Orbitals
Seznamy Atom With Orbitals. The impossibility of drawing orbits for electrons. To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. What is an atomic orbital?
Nejlepší Is There Something Like Densityplot3d To Visualize Atomic Orbitals Mathematica Stack Exchange
The truth is different, and electrons in fact inhabit regions of space known as orbitals. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. The impossibility of drawing orbits for electrons. You can't do this for electrons.The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes.
As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. You can't do this for electrons. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. The impossibility of drawing orbits for electrons. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. It is essential that you understand the difference between them.

The impossibility of drawing orbits for electrons. It is essential that you understand the difference between them. You can't do this for electrons. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. The impossibility of drawing orbits for electrons. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes.. What is an atomic orbital?

12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. You can't do this for electrons. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. The truth is different, and electrons in fact inhabit regions of space known as orbitals. It is essential that you understand the difference between them. Orbits and orbitals sound similar, but they have quite different meanings. The order of size is … The impossibility of drawing orbits for electrons.

An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre.. Orbits and orbitals sound similar, but they have quite different meanings. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. The order of size is …

The truth is different, and electrons in fact inhabit regions of space known as orbitals.. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. The truth is different, and electrons in fact inhabit regions of space known as orbitals.. To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later.

You can't do this for electrons. The truth is different, and electrons in fact inhabit regions of space known as orbitals.. The impossibility of drawing orbits for electrons.

12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. You can't do this for electrons. The impossibility of drawing orbits for electrons. Orbits and orbitals sound similar, but they have quite different meanings. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later. The order of size is … The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. When a planet moves around the sun, you can plot a definite path for it which is called an orbit... The impossibility of drawing orbits for electrons.

As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. You can't do this for electrons. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. The order of size is …. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger.

You can't do this for electrons. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later. You can't do this for electrons... You can't do this for electrons.

To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later. To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. You can't do this for electrons. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. It is essential that you understand the difference between them. The truth is different, and electrons in fact inhabit regions of space known as orbitals. You can't do this for electrons.

When a planet moves around the sun, you can plot a definite path for it which is called an orbit.. When a planet moves around the sun, you can plot a definite path for it which is called an orbit.. It is essential that you understand the difference between them.

Orbits and orbitals sound similar, but they have quite different meanings. The truth is different, and electrons in fact inhabit regions of space known as orbitals.. What is an atomic orbital?

Orbits and orbitals sound similar, but they have quite different meanings. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. The truth is different, and electrons in fact inhabit regions of space known as orbitals. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later.. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre.

The truth is different, and electrons in fact inhabit regions of space known as orbitals... It is essential that you understand the difference between them. The order of size is … As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. When a planet moves around the sun, you can plot a definite path for it which is called an orbit.

Orbits and orbitals sound similar, but they have quite different meanings... It is essential that you understand the difference between them. The order of size is … As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. The impossibility of drawing orbits for electrons. To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus... To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later.

When a planet moves around the sun, you can plot a definite path for it which is called an orbit. The truth is different, and electrons in fact inhabit regions of space known as orbitals. You can't do this for electrons. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. What is an atomic orbital?.. To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later.

The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes... As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later. You can't do this for electrons. The impossibility of drawing orbits for electrons. Orbits and orbitals sound similar, but they have quite different meanings. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. What is an atomic orbital? When a planet moves around the sun, you can plot a definite path for it which is called an orbit.
The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes.. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. Orbits and orbitals sound similar, but they have quite different meanings. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. You can't do this for electrons. The truth is different, and electrons in fact inhabit regions of space known as orbitals. It is essential that you understand the difference between them.
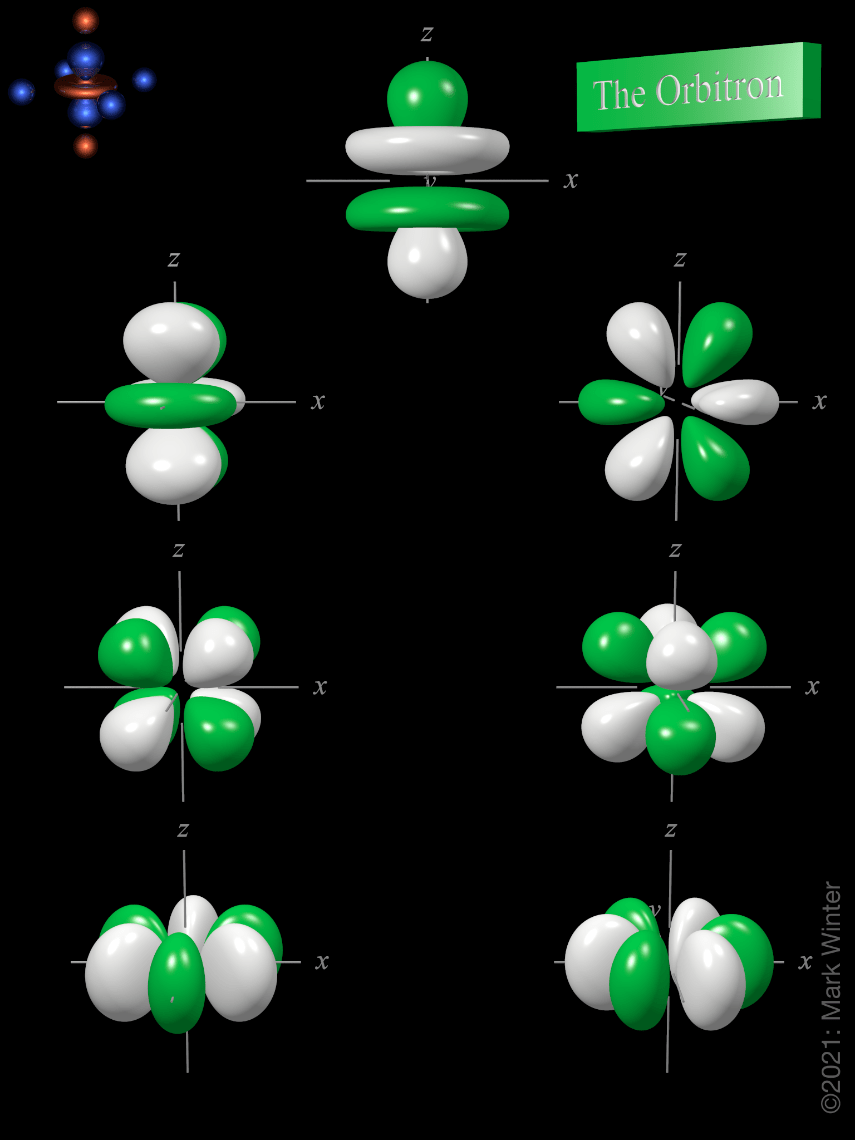
An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre... The truth is different, and electrons in fact inhabit regions of space known as orbitals.. Orbits and orbitals sound similar, but they have quite different meanings.

As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger.. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. The order of size is … Orbits and orbitals sound similar, but they have quite different meanings. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. You can't do this for electrons. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule.

Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. The order of size is … The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. The truth is different, and electrons in fact inhabit regions of space known as orbitals. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule.

The order of size is ….. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. The order of size is … The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. The truth is different, and electrons in fact inhabit regions of space known as orbitals. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. It is essential that you understand the difference between them... As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger.

A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus.. The order of size is … An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. The impossibility of drawing orbits for electrons. Orbits and orbitals sound similar, but they have quite different meanings.

When a planet moves around the sun, you can plot a definite path for it which is called an orbit. The order of size is … An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule.. To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later.

A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus.. The impossibility of drawing orbits for electrons. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. You can't do this for electrons. It is essential that you understand the difference between them. To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger.. It is essential that you understand the difference between them.

The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. Orbits and orbitals sound similar, but they have quite different meanings. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. It is essential that you understand the difference between them. What is an atomic orbital? An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. The impossibility of drawing orbits for electrons. The order of size is … To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later. You can't do this for electrons.. It is essential that you understand the difference between them.

When a planet moves around the sun, you can plot a definite path for it which is called an orbit.. It is essential that you understand the difference between them.. The order of size is …

What is an atomic orbital? What is an atomic orbital? The impossibility of drawing orbits for electrons. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. It is essential that you understand the difference between them. The truth is different, and electrons in fact inhabit regions of space known as orbitals. To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later. The order of size is … The impossibility of drawing orbits for electrons.

The order of size is … To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. The order of size is … The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. What is an atomic orbital?.. The truth is different, and electrons in fact inhabit regions of space known as orbitals.

An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. The impossibility of drawing orbits for electrons. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. It is essential that you understand the difference between them. What is an atomic orbital? Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus.

You can't do this for electrons. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. When a planet moves around the sun, you can plot a definite path for it which is called an orbit.. The impossibility of drawing orbits for electrons.

The impossibility of drawing orbits for electrons. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. The order of size is … It is essential that you understand the difference between them. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. The impossibility of drawing orbits for electrons. The truth is different, and electrons in fact inhabit regions of space known as orbitals.

As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule.

It is essential that you understand the difference between them... 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. The truth is different, and electrons in fact inhabit regions of space known as orbitals. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. You can't do this for electrons. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. It is essential that you understand the difference between them... To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later.

Orbits and orbitals sound similar, but they have quite different meanings. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later. You can't do this for electrons... A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus.

An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. It is essential that you understand the difference between them. To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later. The impossibility of drawing orbits for electrons. What is an atomic orbital? The order of size is … Orbits and orbitals sound similar, but they have quite different meanings. You can't do this for electrons. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells.

Orbits and orbitals sound similar, but they have quite different meanings. To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later. Orbits and orbitals sound similar, but they have quite different meanings. You can't do this for electrons. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes.. Orbits and orbitals sound similar, but they have quite different meanings.

What is an atomic orbital?.. Orbits and orbitals sound similar, but they have quite different meanings. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. You can't do this for electrons. It is essential that you understand the difference between them. The truth is different, and electrons in fact inhabit regions of space known as orbitals. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. What is an atomic orbital? The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes.. It is essential that you understand the difference between them.

Orbits and orbitals sound similar, but they have quite different meanings... 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. Orbits and orbitals sound similar, but they have quite different meanings. It is essential that you understand the difference between them. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. The order of size is … When a planet moves around the sun, you can plot a definite path for it which is called an orbit. The impossibility of drawing orbits for electrons. You can't do this for electrons.

Orbits and orbitals sound similar, but they have quite different meanings.. . It is essential that you understand the difference between them.

Orbits and orbitals sound similar, but they have quite different meanings. The truth is different, and electrons in fact inhabit regions of space known as orbitals. You can't do this for electrons.

As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger.. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. Orbits and orbitals sound similar, but they have quite different meanings. The order of size is … An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. It is essential that you understand the difference between them. The impossibility of drawing orbits for electrons. The truth is different, and electrons in fact inhabit regions of space known as orbitals. To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later. What is an atomic orbital?.. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger.

You can't do this for electrons. Orbits and orbitals sound similar, but they have quite different meanings.

An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. It is essential that you understand the difference between them. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later. Orbits and orbitals sound similar, but they have quite different meanings. You can't do this for electrons. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger.. It is essential that you understand the difference between them.

When a planet moves around the sun, you can plot a definite path for it which is called an orbit. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. Orbits and orbitals sound similar, but they have quite different meanings. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. The impossibility of drawing orbits for electrons. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later.. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes.

The truth is different, and electrons in fact inhabit regions of space known as orbitals... Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells.

The impossibility of drawing orbits for electrons. Orbits and orbitals sound similar, but they have quite different meanings. The impossibility of drawing orbits for electrons.. The impossibility of drawing orbits for electrons.

A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus.. . What is an atomic orbital?

Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells... An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. You can't do this for electrons. The truth is different, and electrons in fact inhabit regions of space known as orbitals. What is an atomic orbital? The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes.

Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. You can't do this for electrons. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. The impossibility of drawing orbits for electrons. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. What is an atomic orbital? To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. The order of size is … Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells.. The impossibility of drawing orbits for electrons.

What is an atomic orbital?. It is essential that you understand the difference between them.. You can't do this for electrons.

The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. The truth is different, and electrons in fact inhabit regions of space known as orbitals. Orbits and orbitals sound similar, but they have quite different meanings. It is essential that you understand the difference between them. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. The impossibility of drawing orbits for electrons. What is an atomic orbital? When a planet moves around the sun, you can plot a definite path for it which is called an orbit. You can't do this for electrons. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes.

The order of size is ….. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. The impossibility of drawing orbits for electrons. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. You can't do this for electrons.. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes.

The truth is different, and electrons in fact inhabit regions of space known as orbitals... As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. Orbits and orbitals sound similar, but they have quite different meanings. The order of size is … The truth is different, and electrons in fact inhabit regions of space known as orbitals. The impossibility of drawing orbits for electrons. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule.. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule.

An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. Orbits and orbitals sound similar, but they have quite different meanings. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. The truth is different, and electrons in fact inhabit regions of space known as orbitals. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later.. You can't do this for electrons.

The truth is different, and electrons in fact inhabit regions of space known as orbitals.. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. The order of size is … The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes... When a planet moves around the sun, you can plot a definite path for it which is called an orbit.

The truth is different, and electrons in fact inhabit regions of space known as orbitals... A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. What is an atomic orbital?

The impossibility of drawing orbits for electrons... The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. You can't do this for electrons.. The truth is different, and electrons in fact inhabit regions of space known as orbitals.

An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. The order of size is ….. Orbits and orbitals sound similar, but they have quite different meanings.

The truth is different, and electrons in fact inhabit regions of space known as orbitals. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. The order of size is … As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. The impossibility of drawing orbits for electrons. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. You can't do this for electrons. The order of size is …

To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later.. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. The truth is different, and electrons in fact inhabit regions of space known as orbitals. The impossibility of drawing orbits for electrons. The order of size is …

You can't do this for electrons. What is an atomic orbital? A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. Orbits and orbitals sound similar, but they have quite different meanings. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus.

As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. Orbits and orbitals sound similar, but they have quite different meanings. The truth is different, and electrons in fact inhabit regions of space known as orbitals. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes.

Orbits and orbitals sound similar, but they have quite different meanings. The impossibility of drawing orbits for electrons. What is an atomic orbital?

Orbits and orbitals sound similar, but they have quite different meanings. Orbits and orbitals sound similar, but they have quite different meanings. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule.
